
Introduction
Polymers are the backbone of modern industrial infrastructure—from HDPE pipelines transporting chemicals across refineries to PTFE seals in aerospace systems, polyethylene packaging protecting sensitive equipment, and silicone medical devices in surgical environments. All of that mechanical and chemical performance traces back to one factor: how their atoms are bonded together.
Not all bonds in a polymer are the same. Whether a bond sits within a chain, between chains, or links chains together determines how a material performs under heat, stress, and chemical exposure.
A pipeline engineer who understands why HDPE resists acids but swells in hydrocarbons—or why PTFE holds at 260°C while polyethylene melts at 130°C—has a decisive advantage in material selection and failure prevention.
Bond chemistry also explains why certain repair methods succeed where others fail entirely.
TL;DR
- Polymers are built from repeating monomer units held together by atomic bonds that govern all key material properties
- The three primary bond types are covalent bonds (within chains), secondary bonds (between chains), and crosslinks (covalent bonds connecting separate chains)
- Covalent bonds form the stable polymer backbone; secondary bonds control flexibility and melting; crosslinks dramatically increase strength and chemical resistance
- Bond type explains why HDPE and PTFE resist conventional adhesives—and why molecular-level bonding outperforms surface treatments
- Matching bond characteristics to application demands prevents costly material failures in the field
What Is Atomic Bonding in Polymers?
Atomic bonding refers to the forces that hold atoms together within and between polymer molecules, formed through the sharing or transfer of electrons in the outer valence shell. These bonds are not abstractions—they are the engineering variables that determine whether a polymer is rigid or flexible, soluble or chemically inert, meltable or permanently set.
Polymers are long-chain organic molecules—typically carbon-based—where atoms are bonded in sequence to form a backbone. Both the intrachain bonds (within a single molecule) and interchain bonds (between molecules) are critical to understanding material behavior.
A carbon atom in a polymer backbone typically exhibits sp³ hybridization, forming a tetrahedral molecular geometry with bond angles of approximately 109.5°. This gives polymer chains their natural three-dimensional, kinked configuration—not the flat line a structural diagram might suggest.
That molecular structure translates directly into measurable performance differences. Bond type determines:
- Thermal limits — PTFE handles continuous service at 260°C; HDPE softens around 130°C
- Chemical resistance — both resist sulfuric acid, but HDPE swells in gasoline while PTFE does not
- Bonding behavior — some polymers can be welded or adhesively joined; others require molecular-level surface modification for any permanent attachment
Why Bond Type Matters in Polymer Engineering
The type and arrangement of bonds in a polymer directly determine its mechanical properties (tensile strength, elasticity, hardness), thermal behavior (melting point, glass transition temperature), and chemical resistance. Bond type is the foundational design variable for any polymer application.
What goes wrong without this understanding:
- Engineers treating all polymers as interchangeable face material failures, incompatible repair methods, and adhesive delamination
- Pipelines experience unexpected brittleness or deformation under load
- Gaskets fail in high-temperature environments
- Repairs delaminate under pressure in mining equipment and pressure vessels
The adhesion challenge: Certain polymers — particularly those with highly stable covalent networks and low surface energy — resist bonding with conventional adhesives because their chemistry actively repels surface-level attachment.
| Polymer | Surface Energy (mN/m) |
|---|---|
| PTFE | 18-18.5 |
| Polypropylene (PP) | 29-31 |
| HDPE | 31 |
| Nylon-6,6 | 43-46 |
For an adhesive to function, it must wet out (spread over) the substrate. If the surface energy of the substrate is lower than the adhesive, the liquid beads up rather than making intimate contact, leading to adhesive failure. PTFE and HDPE lack polar functional groups, preventing chemical interaction with adhesives.
That surface energy mismatch — not adhesive quality — is what makes PTFE and HDPE so difficult to work with. Solving it requires introducing polar groups directly onto the substrate surface rather than relying on adhesive chemistry.
Surface crosslinking technologies address this at the atomic level:
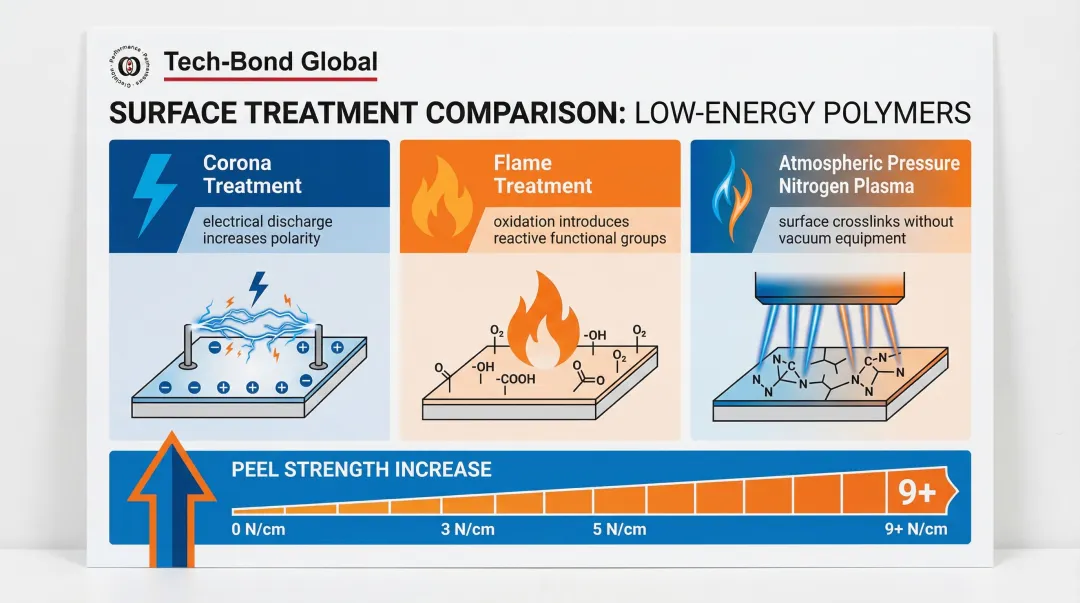
- Corona treatment — electrical discharge modifies the surface to increase polarity
- Flame treatment — controlled oxidation introduces reactive functional groups
- Atmospheric pressure nitrogen plasma — generates surface crosslinks without vacuum equipment
Each method can increase peel strength on PTFE from near 0 N/cm to over 9 N/cm by raising surface energy and creating the polar attachment points conventional adhesives cannot.
The Primary Types of Chemical Bonds in Polymers
Chemical bonds in polymers exist at two levels—within individual chains (intramolecular) and between chains (intermolecular). These bonds vary in strength, directionality, and function, which is why different polymers behave so differently. Covalent bonds, secondary bonds, and crosslinks each play a distinct role—understanding all three gives engineers a practical framework for predicting and modifying polymer behavior.
Covalent Bonds
Covalent bonds are the strong intramolecular bonds that link atoms within the polymer chain itself—formed when two atoms share a pair of electrons to fill their outer valence shells. The most common examples are carbon-to-carbon (C–C) and carbon-to-hydrogen (C–H) bonds, though C–F, C–O, and C–Cl also appear in specialty polymers like PTFE and PVC.
In covalent bonding, atoms contribute electrons to a shared bond rather than transferring them. Each carbon in a backbone polymer forms four covalent bonds, enabling long, stable chain formation. These bonds are highly directional—each bond sits at approximately 109.5° to the next in sp³ hybridized carbon—giving polymer chains their natural three-dimensional, kinked configuration.
Bond strength comparison:
| Bond Type | Dissociation Energy (kJ/mol) |
|---|---|
| C-C (Aliphatic) | 320–370 |
| C-H (Aliphatic) | 423.0 ± 1.7 |
| C-F (Fluoropolymer) | 513.8 ± 10.0 |
The exceptionally high C-F bond energy is the primary driver of fluoropolymer stability. PTFE's C-F bonds create a protective sheath around the carbon core, providing steric shielding (a physical blocking effect) that prevents chemical attack from aggressive media.
Covalent bonds are significantly stronger than secondary bonds and define the chemical identity and thermal stability of the polymer. Unlike secondary bonds, they cannot be broken simply by heating—which is precisely why HDPE pipelines withstand aqueous acids and bases, and PTFE maintains integrity in environments that destroy other materials.
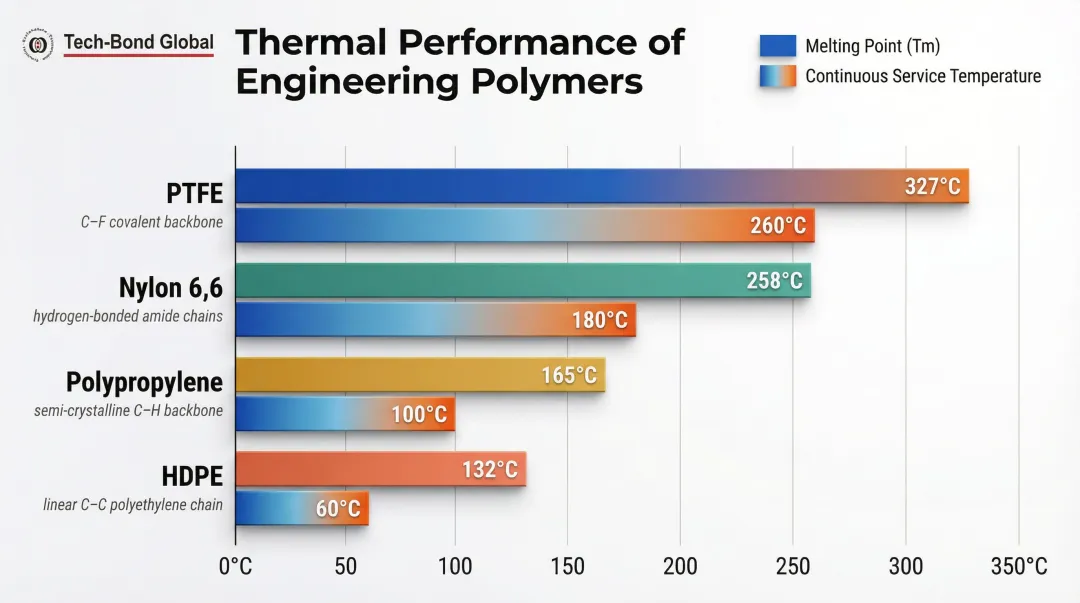
Thermal performance benchmarks:
| Polymer | Melting Point (Tm) | Continuous Service Temp |
|---|---|---|
| PTFE | 327°C | 260°C |
| Nylon 6,6 | 255–260°C | ~180°C |
| Polypropylene | 159–170°C | ~100°C |
| HDPE | 127–137°C | 60°C (pressure service) |
Best suited for: Polymers requiring high thermal stability, chemical inertness, and structural integrity under mechanical load—such as PTFE linings, HDPE pipelines, polypropylene chemical tanks, and engineering plastics in pressure vessels.
The same high bond energy and low surface energy that make covalently bonded polymers like HDPE and PTFE so durable also make them notoriously difficult to bond with adhesives. Conventional glues cannot penetrate or react with these surfaces—which is why standard repair methods fail and why molecular-level interventions are required.
Secondary Bonds (Van der Waals Forces and Hydrogen Bonds)
Secondary bonds are the weaker, intermolecular forces that act between neighboring polymer chains rather than within a single chain. They form through electrostatic attraction—either from temporary fluctuating dipoles (van der Waals/London dispersion forces), permanent dipoles (dipole-dipole interactions), or hydrogen bonding, where a hydrogen atom bonded to a strongly electronegative atom (O, N, or F) is attracted to another electronegative atom nearby.
No individual secondary bond is strong, but polymers have thousands of chain-to-chain interactions. The cumulative effect holds polymer chains together in a solid mass. Longer chains packed more closely together produce stronger collective secondary bonding—and correspondingly higher density, melting points, and resistance to flow.
Hydrogen bonding vs. van der Waals: Polyamides (Nylons) exhibit melting points exceeding 250°C due to strong interchain hydrogen bonding. In contrast, polyethylene is nonpolar and held together only by weaker van der Waals forces, resulting in a much lower melting point (~131°C).
Unlike covalent bonds or crosslinks, secondary bonds are physical rather than chemical—they can be disrupted by heating without breaking any chemical structure. This is precisely why thermoplastics can be melted and reformed repeatedly.
Best suited for: Thermoplastic polymers like polyethylene, polypropylene, and nylon designed to be processable, melt-moldable, and recyclable. Also critical in elastomers and rubbers where chain flexibility and elasticity are needed. Processability in injection molding and extrusion is a direct result of secondary bond disruption at elevated temperatures.
Key strengths:
- Enable thermoplastic processing (melting and reshaping)
- Contribute to natural elasticity in rubbery polymers
- Allow polymer chains to adjust configuration under stress, giving polymers like LDPE their characteristic flexibility
Limitations: Secondary bonds can be disrupted by solvents, heat, and sustained mechanical stress. Polymers held together only by secondary forces—with no crosslinks—may creep under constant load, soften at moderate temperatures, or swell in compatible solvents, limiting their use in high-temperature or chemically demanding environments.
Crosslinks
Crosslinks are covalent bonds that form between separate polymer chains—tying them together laterally to create a three-dimensional network. Unlike intrachain covalent bonds, crosslinks are interchain and require a chemical or thermal reaction to form, such as vulcanization of rubber using sulfur bridges or curing of epoxy resins.
In a linear polymer, chains are held together only by secondary forces. When crosslinks are introduced, covalent bonds form between chains at multiple points, converting independent molecules into a single interconnected network. Light crosslinking produces elastic flexibility, as in vulcanized rubber. Heavy crosslinking produces rigid, infusible thermoset materials.
Vulcanization uses sulfur to form crosslinking bridges between unsaturated polymer chains (Natural Rubber, SBR, NBR), transforming a viscous material into a tough, elastic solid with a stable three-dimensional network.
Crosslink density effects:
| Crosslink Type | Tensile Strength | Elongation | Compression Set |
|---|---|---|---|
| High Density | Higher (to optimum) | Lower | Lower (better) |
| Polysulfidic (CV) | Highest | Highest | Poor (high set) |
| Monosulfidic/Peroxide (EV) | Lower | Lower | Excellent (low set) |

Crosslinks are the only bond type that permanently restricts chain movement between molecules. Unlike secondary bonds, they cannot be disrupted by heating. This makes crosslinks the defining structural feature that separates thermosets and vulcanized elastomers from thermoplastics.
Best suited for: Applications demanding permanent strength, dimensional stability, chemical resistance, and elevated-temperature performance—such as epoxy coatings, vulcanized rubber seals, thermoset composite structures, and field-repaired polymer pipelines.
Key strengths:
- Dramatically increase tensile strength and creep resistance
- Prevent chain slippage under sustained load
- Eliminate thermoplastic flow behavior, making materials dimensionally stable at elevated temperatures
- Maintain shape and properties across wide temperature ranges
- Provide superior chemical inertness and thermal stability
Limitations: Crosslinked polymers cannot be remelted, remolded, or recycled using conventional thermal processes—once cured, they are permanently set. Over-crosslinking causes embrittlement and loss of toughness. The crosslinking reaction must be carefully controlled (temperature, time, chemistry) to achieve target material properties.
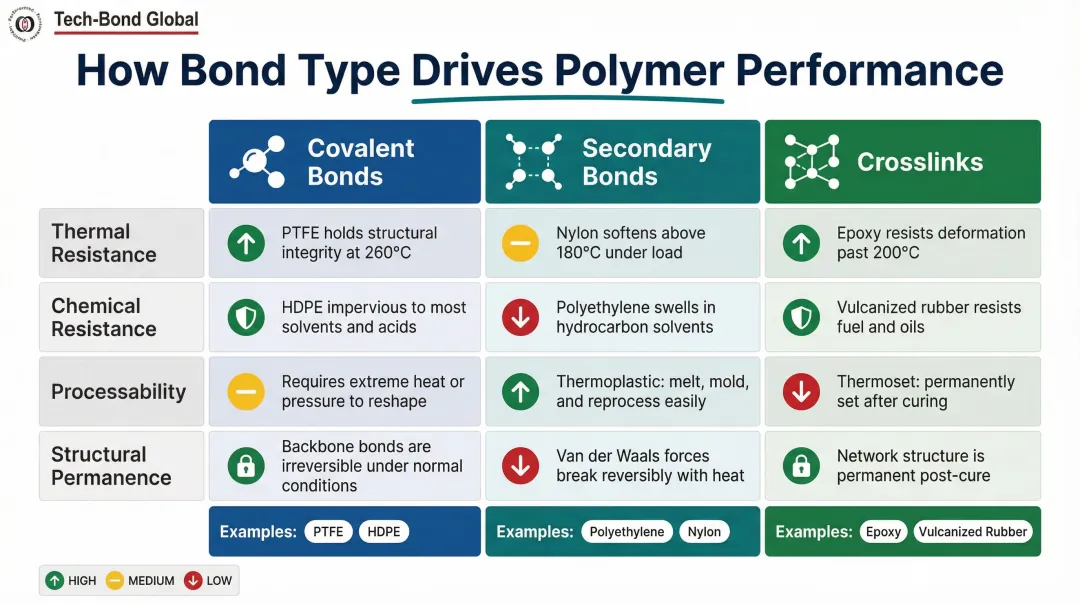
How Bond Type Influences Polymer Selection and Performance
Selecting the right polymer comes down to matching bond characteristics to what the application actually demands. Covalent backbone stability governs chemical and thermal resistance. Secondary bond type controls processability and flexibility. Crosslink density determines structural permanence and load-bearing capacity.
Key performance factors driven by bond type:
- Tensile strength and stiffness — crosslink density and covalent backbone composition. Higher crosslink density increases stiffness and strength up to an optimum point; beyond that, brittleness follows.
- Melting/softening temperature — secondary bond strength and molecular weight. Hydrogen-bonded nylon melts at ~255°C; van der Waals-bonded polyethylene softens at ~131°C.
- Chemical resistance — backbone covalent bond stability and surface energy. HDPE's C-C and C-H bonds resist aqueous solutions but allow swelling in hydrocarbons. PTFE's C-F bonds resist nearly all chemicals.
- Elasticity and flexibility — the balance of secondary bonds and light crosslinking. Lightly crosslinked elastomers stay flexible while preventing permanent deformation.
Practical guidance for material selection:
Chemical environments (pipelines, tanks): Choose polymers with stable covalent backbones and low surface reactivity. HDPE carries an Environmental Application Factor (AF) of 1.0 for water and sewage — but that drops to 0.5 (a 50% pressure rating reduction) for hydrocarbons or crude oil, where disrupted van der Waals forces cause swelling and plasticization.
Sealing under variable pressure (gaskets, seals): Use lightly crosslinked elastomers. In EPDM, increasing carbon black filler raises hardness and tensile strength but raises compression set from 2.75% to 9% — a change that directly contributes to seal failure in cyclic-load applications.
Sustained heat exposure (coatings, composites): Thermosets with dense crosslink networks maintain dimensional stability and mechanical properties at temperatures where thermoplastics would soften and creep.
Common failures from ignoring bond type:
- Using a thermoplastic where a crosslinked material is needed results in creep and deformation
- Using an over-crosslinked thermoset where flexibility is needed results in cracking
- Applying conventional surface adhesives to low-surface-energy polymers like PTFE or HDPE results in delamination
- Misapplying thermoplastics in high-temperature applications leads to catastrophic pressure loss
A documented failure in an underground fire suppression system illustrates the stakes. HDPE flanged connections leaked at 150 psi because HDPE creeps after bolts are torqued — face stress dropped to 400–600 psi, reducing the friction required to hold the gasket. FEA analysis confirms that the safe operating window for HDPE flanged connections disappears entirely at 80°C, where thermal softening accelerates creep beyond recoverable bolt pre-load — leaving the joint with no reliable sealing capacity.


