
Introduction
Bond failures in critical infrastructure don't announce themselves—they delaminate under pressure, fail during thermal cycles, and compromise structural integrity long after the repair crew has left. Engineers and asset managers across pipeline, mining, marine, and industrial repair sectors encounter these failures regularly, often without a clear explanation for why.
The root cause traces back to one consistently overlooked fact: metal-polymer adhesion is a molecular phenomenon governed by interfacial chemistry, not simply surface contact.
Despite appearing in countless materials specifications and repair discussions, the mechanistic side of metal-polymer adhesion rarely gets explained. What follows covers how the bonding process works, what drives it, and what separates a bond that lasts decades from one that fails under the first thermal cycle.
TL;DR
- Metal-polymer adhesion requires more than contact—interfacial chemistry determines durability or failure.
- Five mechanisms drive bonding: Van der Waals forces, acid-base interactions, hydrogen bonding, covalent coupling, and mechanical interlocking.
- Low-surface-energy polymers (HDPE, PTFE, polypropylene) resist conventional adhesives without surface activation.
- Moisture attacks the interface 450 times faster than through bulk polymer, while thermal cycling accelerates delamination.
- Surface-level molecular crosslinking outperforms adhesive films in harsh, high-stress applications.
What Is Metal-Polymer Adhesion?
Metal-polymer adhesion is the establishment of stable interfacial forces or bonds between a polymer material and a metallic substrate. These bonds form through a combination of physical interactions and chemical reactions at the nanoscale interface — not through bulk material properties alone.
The Fundamental Incompatibility
Metals and polymers are fundamentally incompatible at the surface level. Metals are inorganic, crystalline solids with high surface energy (700–1,650 mJ/m² for common structural metals) and native oxide layers. Polymers like HDPE, polypropylene, and PTFE are organic, often amorphous or semi-crystalline, with surface energies of just 18–33 mJ/m² — hundreds of times lower than metals (see Forgeway's surface energy reference chart).
This massive surface energy mismatch means polymers lack inherent affinity for metallic surfaces. Bridging this gap requires deliberate molecular engineering.
Adhesion vs. Adhesive Bonding
These terms are often confused, but they describe different things:
- Adhesion refers to the molecular phenomenon of interfacial force formation — the physics and chemistry occurring at the contact zone between two materials.
- Adhesive bonding is the engineering application using a third material (glue, epoxy, primer) to facilitate that adhesion.
Confusing the two leads to poor design and repair decisions. The bonding mechanism at work in your joint — mechanical interlocking, chemical reaction, or molecular crosslinking — directly determines the repair strategy that will actually hold under load and environmental stress.
How Metal-Polymer Adhesion Works at the Molecular Level
When a polymer contacts a metal surface, the first event is wetting—the polymer must achieve intimate molecular contact with the metal (or its oxide layer) before any bonding forces can act. The quality of this contact determines the maximum achievable bond strength.
Physical Forces and Surface Wetting
Van der Waals (dispersion) forces are the baseline of all polymer-metal contact. These weak physical forces exist whenever molecules are in proximity, but alone they cannot sustain structural loads, particularly under environmental stress. They are necessary but not sufficient for durable adhesion.
Surface wettability depends on surface energy matching. The polymer's surface energy must be close to or lower than the metal's surface energy for wetting to occur. Metals generally have high surface energy, but oxidation and contamination reduce it. That reduction is why surface preparation affects bond performance, not just surface appearance.
Chemical Bonding at the Interface
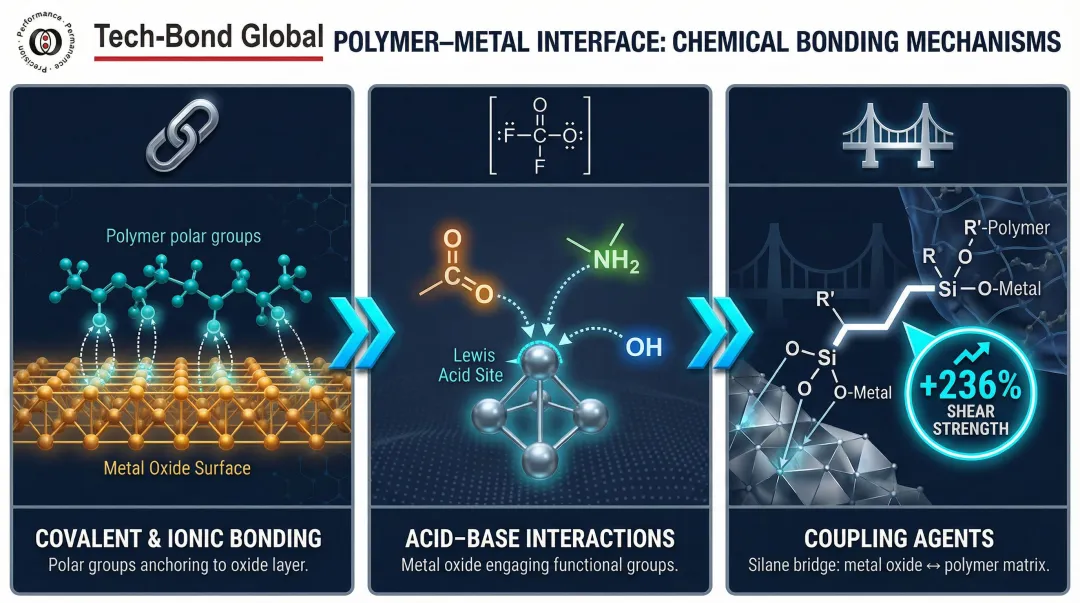
Three bonding mechanisms drive durable polymer-metal adhesion at the molecular level:
- Covalent and ionic bonding: The metal oxide layer on most structural metals forms covalent and coordinate bonds with polar functional groups on the polymer. These bonds are substantially stronger than physical forces and form the basis of durable adhesion.
- Acid-base interactions (Lewis and Brønsted): Metal oxides act as Lewis acid or base sites; polymer functional groups (carbonyl, amine, hydroxyl) interact with these sites to form strong interfacial bonds. Surface chemistry of both materials—not just the adhesive—determines joint durability.
- Coupling agents: Organofunctional silanes and titanates act as molecular bridges, bonding chemically to the metal oxide on one end and reacting with the polymer matrix on the other. Silane KH550, for example, increased stainless steel/epoxy shear strength from 6.05 MPa to 20.36 MPa—a 236% improvement through chemical bridging.
Mechanical Interlocking
Surface roughness contributes through mechanical interlocking. Micro and nano-scale features increase real contact area and allow the polymer to penetrate those features during application, creating physical anchoring.
Abrasive blasting mild steel surfaces increased shear strength by 60% compared to non-abraded surfaces. Mechanical interlocking and chemical adhesion are complementary: roughness improves wetting and contact area, but chemical bonding carries the load under peel, vibration, or cyclic stress.
Engineering Approaches to Reliable Metal-Polymer Bonding
Surface Preparation: The Most Critical Step
Surface preparation of the metal is the most critical, most frequently neglected engineering step. Contamination (oils, oxides, process residues) prevents intimate molecular contact and renders even the best adhesive ineffective.
Three preparation approaches restore high-energy, chemically reactive surfaces:
- Mechanical: Abrasive blasting, grinding
- Chemical: Acid etching, solvent cleaning
- Plasma/UV: Advanced surface activation
ASTM D2651 provides standard guidance for preparation of metal surfaces for adhesive bonding.
Adhesion Promoters and Primers
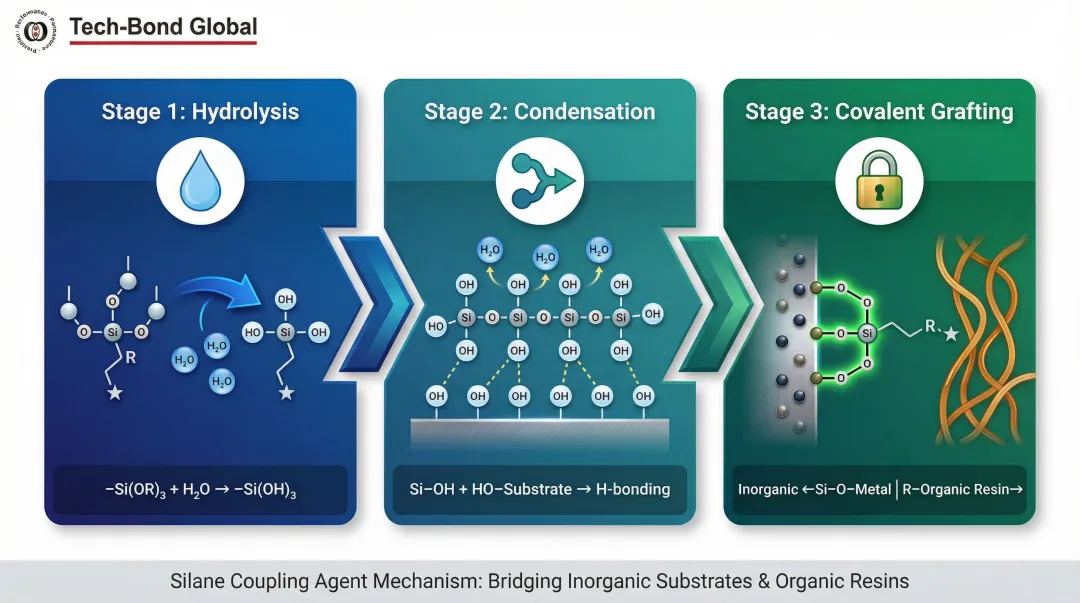
Silane coupling agents, organotitanates, and reactive primers are applied to the metal surface to convert it from an inorganic to a hybrid organic-inorganic interface. These molecules self-assemble on the oxide surface and then react with the incoming polymer layer.
The mechanism involves:
- Hydrolysis: The alkoxy group reacts with water to form silanol (Si-OH)
- Condensation: Silanols form oligomers and hydrogen bond with substrate hydroxyl groups
- Covalent grafting: During curing, a covalent oxane bond (Si-O-Metal) forms with the inorganic substrate while the organofunctional group attaches to the organic resin
Titanate coupling agents similarly improve bonding, with one study showing adhesive strength between phenolic adhesive and stainless steel increased from 26.21 MPa to 39.56 MPa at optimal titanate concentration.
Polymer Selection and Formulation
The surface energy, polarity, and functional group density of the polymer dictate which bonding mechanisms are available. Semicrystalline non-polar polymers (HDPE, PTFE, polypropylene) have few functional groups for interfacial bonding and require surface activation before adhesion is achievable.
Surface activation techniques include:
- Plasma treatment: Reduced HDPE contact angle from 79° to 29°, dramatically increasing surface energy
- Corona discharge: Introduces polar functional groups
- Chemical etching: Exposes fresh material and increases surface area
Plasma treatment of PTFE reduced contact angle from 116° to 13°, enabling bonding where none was previously possible.
Molecular Crosslinking Approaches
The most durable engineering approach is molecular crosslinking. Rather than relying on an adhesive film between polymer and metal, this approach activates and crosslinks the polymer surface itself, creating direct covalent bonds at the interface.
Tech-Bond Global's patented Polymer Bonding Process operates on this principle. The system uses three components working in sequence:
- Poly Prep: Permanently changes surface energy by etching the polymer to expose fresh material and create bonding sites
- SI Blue cyanoacrylate adhesive: Available in viscosities from 0–5 cP for wicking applications to 1,500 cP for vertical surfaces
- Accelerator catalyst: Triggers the crosslinking reaction that drives permanent molecular-level bonds

Together, these components bond polymers to metal substrates — including materials historically considered unbondable such as HDPE, PTFE, and polypropylene — without conventional adhesives.
The Accelerator triggers an exothermic chemical reaction that drives crosslinking at approximately 117-120°F (47-49°C), resulting in bonds that resist stress, heat, weathering, moisture, and most chemicals.
Process Control Parameters
Inconsistency in any of these factors produces adhesion variability even with the same materials and system:
- Temperature and pressure during application
- Cure time and environment (humidity, UV exposure)
- Surface preparation window (time between preparation and application)
- Post-bond conditioning
Key Factors That Affect Metal-Polymer Adhesion Performance
Primary Variables
Bond performance in industrial environments depends on several interacting factors:
- Polymer surface energy, crystallinity, and polar group density
- Metal oxide layer type — aluminum oxide, iron oxide, and zinc oxide each respond differently to coupling agents
- Surface cleanliness and roughness profile, beyond which additional roughening yields diminishing strength returns
- Bonding method: adhesive bonding versus molecular-level crosslinking
Environmental Threats to Bond Integrity
Moisture is the most damaging threat. Industry sources report that water diffuses up to 450 times faster along the interface than through the polymer bulk, hydrolyzing interfacial bonds. Water reaching the interface forms an electrolyte macroscopic phase, facilitating cathodic delamination.
Thermal cycling compounds the problem through differential expansion. Polymers expand 2–20 times more than metals, generating cyclic stress at the interface that accelerates delamination over time:
| Material | Linear CTE (µm/m-°C) | Ratio vs. Steel |
|---|---|---|
| Steel | 12 | 1.0x |
| Aluminum | 23.6 | 2.0x |
| Epoxy | 45–65 | 3.75–5.42x |
| HDPE | 108–200 | 9.00–16.67x |
| PTFE | 112–135 | 9.33–11.25x |

Standards and Compliance
These degradation mechanisms are precisely why standardized testing exists. Key standards governing metal-polymer bond qualification include:
- ASTM D4541: Pull-off strength of coatings from metal substrates
- ASTM D1002: Apparent shear strength of single-lap-joint adhesively bonded metal specimens
- ISO 24817: Composite repairs for pipework—qualification and design
- ASME PCC-2: Repair of pressure equipment and piping
Operators specifying repair systems for critical infrastructure should verify that bonding solutions have been tested against applicable standards — particularly ASTM D4541 for field-applied coatings and ISO 24817 for composite pipe repairs.
Common Misconceptions and Misapplications in Metal-Polymer Bonding
"More Adhesive = Stronger Bond"
The most damaging misconception: that increasing adhesive quantity improves bond strength. In reality, bond strength decreases quasi-linearly as adhesive thickness increases beyond an optimal threshold (typically 0.1–0.3 mm). Thicker bondlines contain more defects (voids, microcracks) and exhibit higher interface peel stresses.
Durability is determined by interface preparation and chemistry — volume works against you past that optimal range.
"All Polymers Behave the Same"
Low-surface-energy polymers (HDPE, PTFE, silicone rubber, polypropylene) cannot be bonded through standard adhesive application because they lack the surface energy and functional group density to form interfacial bonds.
The lap shear data confirms this: untreated HDPE bonded with epoxy yields only 0.70 MPa, while polyurethane performs worse at 0.46 MPa. Teams that apply standard epoxies or urethanes to these substrates and expect structural adhesion are engineering a failure.
When Bonding Is Unnecessary
The opposite error is just as costly. Molecular bonding approaches are misapplied in:
- Low-stress, non-environmental applications where pressure-sensitive adhesives or mechanical fasteners are adequate
- Environments without moisture, chemical exposure, or thermal cycling
- Situations where teams reach for bonding solutions by default rather than specifying them based on load, environment, and substrate chemistry
Conclusion
Metal-polymer adhesion is ultimately controlled by interface chemistry and molecular-scale interactions. Van der Waals forces, acid-base coupling, covalent bonding, and mechanical interlocking combine at the interface, but no single mechanism is sufficient for demanding industrial service conditions.
The gap between "applying an adhesive" and "engineering a molecular bond" is where most failures originate. For pipeline, mining, and energy applications, knowing which mechanism governs a joint is not academic: it determines whether a repair holds for months or for decades.
With global corrosion costs exceeding $2.5 trillion annually, the cost of getting bonding science wrong is measured in infrastructure failures, not just budget overruns. Applying the right molecular approach from the outset is what separates a repair that lasts from one that needs redoing.
Frequently Asked Questions
What is adhesion in polymers?
Adhesion in polymers refers to the molecular-level forces and bonds that form when a polymer contacts another surface — Van der Waals forces, acid-base interactions, hydrogen bonding, and covalent bonds are the primary mechanisms. The polymer's surface energy and chemistry determine which of these are active and how durable the resulting bond will be.
What are polymer adhesives used for in metal bonding?
Polymer adhesives serve a range of functions in metal bonding:
- Joining structural metal components in assemblies
- Sealing and coating metal substrates for corrosion protection
- Repairing metal structures in the field
Applications span automotive assembly, aerospace joints, pipeline repair, and industrial equipment maintenance across nearly every manufacturing and infrastructure sector.
What is the best adhesive for bonding metal?
The right adhesive depends on the substrate metal, the polymer being bonded, and the operating environment. Epoxies are widely used for structural metal bonding, but in high-stress or chemically exposed joints, molecular bonding systems that create covalent crosslinks at the interface consistently outperform conventional adhesive films.
Can metal adhesives fill gaps?
Some adhesive formulations (structural epoxies, polyurethane sealants) can fill small surface gaps and level minor irregularities, but gap-filling is not the same as true adhesion. Bonds formed over gaps concentrate stress at unsupported edges and often fail faster than bonds on well-prepared, intimate-contact surfaces.
Why is it difficult to bond HDPE or PTFE to metal surfaces?
HDPE, PTFE, and similar polyolefins have very low surface energy (18–33 mJ/m²) and no reactive functional groups. Standard adhesives cannot form covalent or strong polar bonds with these materials. Achieving durable metal adhesion requires surface activation (plasma, chemical etching) or a molecular bonding process that crosslinks the polymer surface directly.
What is the difference between adhesive bonding and molecular bonding of polymers to metal?
Adhesive bonding uses a third material (glue, epoxy, primer) as an intermediary between polymer and metal. The bond is only as strong as the weakest adhesive-substrate interface. Molecular bonding crosslinks the polymer surface itself to the substrate at the atomic scale, eliminating the adhesive layer entirely and creating a bond that can exceed the strength of the parent materials.


