
TLDR
- PTFE is a synthetic fluoropolymer built from carbon and fluorine atoms — known for near-universal chemical resistance, extremely low friction, and a working range of −200°C to +260°C
- Accidentally discovered in 1938 by Roy Plunkett at DuPont and commercialized as Teflon in 1945 (now a Chemours trademark)
- Strengths include chemical inertness, thermal stability, and electrical insulation — key weaknesses are creep under sustained load and poor elastic recovery
- Available in virgin PTFE (maximum chemical resistance) and filled grades (glass, bronze, carbon, stainless) for improved mechanical performance
- Major industries: chemical processing, oil and gas, aerospace, medical, semiconductor manufacturing, and industrial repair
What PTFE Is: Definition, Chemistry, and Origin
Polytetrafluoroethylene (PTFE) is a high-molecular-weight fluoropolymer with the chemical formula (C₂F₄)n, consisting entirely of carbon-fluorine bonds. These C–F bonds are among the strongest in organic chemistry, with a bond dissociation energy of approximately 488 kJ/mol. This exceptional bond strength explains PTFE's remarkable stability and resistance to chemical attack, oxidation, and thermal degradation.
The Accidental Discovery
On April 6, 1938, Dr. Roy J. Plunkett, a chemist at E.I. du Pont de Nemours & Company, was attempting to develop a new refrigerant gas when tetrafluoroethylene polymerized inside a pressurized cylinder, leaving a slippery white solid. This accidental find led to the registration of the Teflon trademark in 1945. Today, Teflon is owned by The Chemours Company, established as an independent public company following a spin-off from DuPont on July 1, 2015. Teflon is a brand name; PTFE is the generic material designation.
What Makes PTFE Structurally Unique
The fluorine atoms form a tight, symmetrical "sheath" around the carbon backbone, creating a non-polar, hydrophobic molecule that is essentially inert to nearly all chemical attack. This structure explains why water cannot wet PTFE and why conventional adhesives fail to bond to it — the surface simply will not interact chemically with most substances.
Manufacturing Challenges
PTFE is produced via free-radical polymerization of tetrafluoroethylene using suspension or emulsion methods. Processing it presents real difficulties: its melting point (327°C) exceeds its decomposition temperature, making conventional injection molding impossible. Instead, PTFE requires cold compression molding followed by sintering — a process closer to powder metallurgy than plastics manufacturing.
These manufacturing constraints reflect a deeper reality: PTFE's properties come almost entirely from its molecular structure, not from additives or processing variations. That consistency is a strength, but PTFE's inherent limitations cannot be engineered away in virgin form. Addressing them typically requires:
- Filled grades — adding glass fiber, carbon, or bronze to improve wear resistance
- Composite structures — laminating PTFE with substrates for dimensional stability
- Surface treatments — etching or plasma activation to overcome its near-zero surface energy for bonding applications
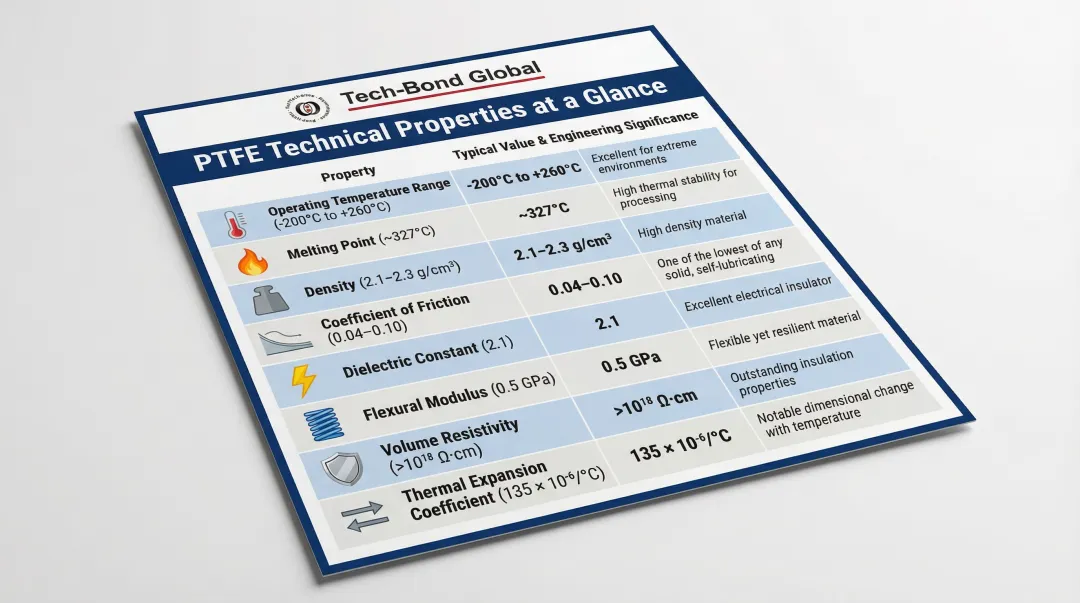
Key Technical Properties of PTFE
PTFE's value comes from a combination of properties that rarely coexist in a single material: chemical universality, broad thermal range, extreme surface inertness, and electrical insulation.
| Property | Typical Value/Range | Engineering Significance |
|---|---|---|
| Operating Temperature | −200°C to +260°C | Maintains stability from cryogenic to extreme heat |
| Melting Point | 327°C | Defines sintering threshold and high-temp limits |
| Density | 2,140–2,200 kg/m³ | High density indicates low porosity in sintered parts |
| Coefficient of Friction | 0.05–0.10 | One of the lowest of any solid material |
| Dielectric Constant | ~2.1 | Excellent electrical insulation across frequencies |
| Young's Modulus | ~0.5 GPa | Relatively soft compared to metals |
| Bulk Resistivity | ≥10¹⁸ Ω·cm | Superior electrical insulator |
| Thermal Expansion | 112–125 × 10⁻⁶ K⁻¹ | Significantly higher than metals; requires tolerance planning |
Chemical Resistance and Inertness
PTFE is resistant to virtually all industrial chemicals — acids, bases, solvents, and oxidizing agents. Only highly reactive metals (alkali metals like sodium and potassium at elevated temperature) and certain fluorinating agents (elemental fluorine, chlorine trifluoride) can attack the carbon-fluorine bonds. This universal resistance makes PTFE the standard material for linings, seals, and gaskets in aggressive chemical environments, codified in standards like ASTM F1545 for plastic-lined ferrous metal pipe.
Thermal Stability and Operating Range
That same C-F bond stability that resists chemical attack also governs PTFE's thermal behavior. The material operates continuously from −200°C to +260°C, covering both cryogenic and high-heat applications. However, degradation begins above 260°C, with pyrolysis occurring above 400°C and releasing toxic fluorocarbon gases and hydrogen fluoride. Engineers must also account for PTFE's high thermal expansion coefficient (112–125 × 10⁻⁶ K⁻¹) — significantly higher than metals — in precision assemblies.
Friction, Surface, and Electrical Properties
PTFE's coefficient of friction (0.05–0.10 against polished steel) is among the lowest of any solid, making it self-lubricating and well-suited for bearings, slide plates, and wear surfaces. No added lubrication is required, which matters in food processing, semiconductor fabrication, and other contamination-sensitive environments.
Its electrical properties follow the same logic: a dielectric constant of ~2.1 and bulk resistivity of 10¹⁸ Ω·cm make it the standard insulating material for high-frequency wiring, coaxial cables, and RF/microwave PCB laminates. Aerospace wiring standards such as SAE AS22759 (M22759) mandate extruded PTFE for severe-environment hookup wires operating up to 260°C.
Mechanical Weaknesses: Creep and Elastic Recovery
PTFE has no elastic memory. Unlike elastomers, it does not crosslink, meaning it permanently deforms (creep or cold flow) under sustained mechanical load. This is a known material characteristic that engineers design around — not an unexpected failure mode.
Virgin PTFE gaskets and seals lose clamping force over time. When subjected to compressive loads in bolted flange joints, PTFE gradually relaxes, leading to decreased residual sealing force and potential leakage. Standard mitigation approaches include:

- Belleville washers (conical disc springs) to maintain bolt load as the gasket relaxes — a "live-loading" technique that adjusts the joint's spring rate
- Controlled bolt torque sequences following ASME PCC-1 guidance, which addresses live tightening as an alternative to start-up retorquing
- Avoiding start-up retorquing on PTFE-based gaskets, which ASME PCC-1 explicitly flags as typically inadvisable
PTFE Grades, Forms, and Composites
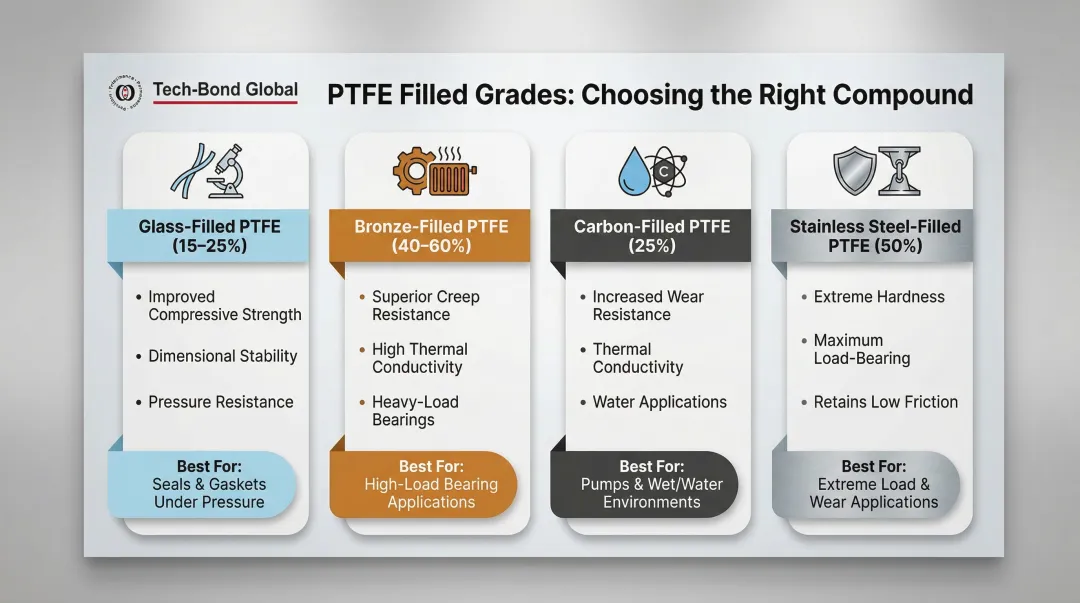
Selecting the correct PTFE grade is as important as selecting PTFE over another material. Two fundamental categories exist: virgin PTFE (unadulterated resin, highest chemical resistance, FDA-compliant) and filled/mechanical PTFE (modified with additives for improved mechanical performance).
Common Filled Grades and What Each Achieves
Filled grades maintain PTFE's chemical and thermal characteristics while addressing its mechanical weaknesses:
- Glass-filled (15–25% glass fiber): Improves compressive strength and reduces deformation under load; enhances dimensional stability and pressure resistance
- Bronze-filled (40–60% bronze): Superior creep resistance and high thermal conductivity; ideal for heavy-load linear bearings
- Carbon-filled (25% carbon): Increases compressive strength, wear resistance, and thermal conductivity; excellent for water applications
- Stainless steel-filled (50% stainless): Extreme hardness and load-bearing capability while retaining low friction
PTFE in Composite and Coated Forms
Beyond bulk material, PTFE appears in several specialized forms:
- Coating: Applied to cookware, industrial rollers, bakeware, and processing equipment for non-stick and chemical-resistant surfaces
- Thin film and ePTFE: Used in composites, medical membranes, and expanded PTFE products like Gore-Tex; ePTFE's microporous structure makes it suitable for cardiovascular grafts and vascular prostheses, with FDA 510(k) clearances covering bypass and dialysis access applications
- PTFE-coated woven glass-fiber fabric: One of the strongest architectural and industrial composites available — the O2 Arena in London's 320-meter diameter canopy is constructed from this material, supported by a tensioned cable net
PTFE-coated E-glass fabric pairs PTFE's chemical imperviousness with E-glass's mechanical reinforcement — a combination that outperforms standard fiberglass wraps in chemical resistance and physical durability. That performance profile is precisely why Tech-Bond Global selected it as the substrate for their Tech Patch Repair System (TPRS), which uses a patented molecular bonding process to create permanent repairs on challenging substrates including HDPE pipe and PTFE-coated materials.
Where PTFE Is Used: Major Industrial Applications
PTFE dominates the fluoropolymer industry, commanding a massive 65.05% share of the global fluoropolymers market in 2024. The global PTFE market was valued at USD 3.63 billion in 2023 and is projected to reach USD 5.25 billion by 2030, representing a CAGR of 5.5%.
That market scale reflects how broadly PTFE's core properties translate into real-world applications — across three distinct performance categories:
- Chemical resistance drives use in pipe linings, valve seats, gaskets, pump components, and reactor vessels
- Low friction makes PTFE the material of choice for bearings, slide plates, gears, bushings, and structural slide bearings for bridges and buildings (specified in standards like EN 1337-2)
- Electrical insulation accounts for roughly 50% of total PTFE production, covering wire and cable insulation, printed circuit boards (including RF/microwave PCB laminates like Rogers RT/duroid 5880), and connector assemblies
Key Sector Applications
- Chemical processing: Reactor vessel linings and pump housings where continuous chemical exposure would degrade most other materials
- Oil and gas: Static seals and gaskets for chemical containment — PEEK or filled PTFE handles high-load dynamic seals better in this environment
- Medical: Cardiovascular grafts, catheters, sutures, and vascular prostheses using FDA-compliant virgin grades
- Food and beverage: Non-stick coatings, conveyor systems, and FDA-approved contact surfaces that require both chemical inertness and easy cleaning
- Aerospace and electronics: Coaxial cables, PCB laminates, and connector assemblies where signal integrity and thermal stability are critical
- Construction: Structural slide bearings, pipe supports, and architectural membranes for long-term load distribution

PTFE Limitations, Operating Boundaries, and Common Misconceptions
Temperature Boundaries and PFOA
PTFE begins degrading at approximately 260°C, with decomposition accelerating above 350°C and pyrolysis occurring above 400°C. At these temperatures, toxic fluorocarbon gases are released, creating inhalation hazards — including "polymer fume fever."
The PFOA question deserves clarity: Perfluorooctanoic acid (PFOA) was historically used as a processing aid in PTFE emulsion polymerization but was never a constituent of the polymer chain itself. In 2006, the U.S. EPA launched the 2010/2015 PFOA Stewardship Program, and participating manufacturers successfully achieved total elimination by 2015, with some reaching elimination as early as 2013. Modern PTFE products are produced without PFOA.
Key Practical Limitations
1. Creep under sustained load: Virgin PTFE is unsuitable for dynamic high-pressure sealing without spring-loaded designs or filled grades.
2. Low wear resistance: Virgin PTFE abrades relatively easily in sliding contact unless filled or backed by metal.
3. Cannot be bonded using conventional adhesives: PTFE's chemically inert, non-polar surface prevents adhesion. Traditional bonding requires aggressive pre-treatment methods:
- Chemical etching: Wet etching using sodium-naphthalene complex dissolved in tetrahydrofuran or diglyme strips fluorine atoms from the surface, leaving a carbonized layer with polar groups that improve wettability
- Plasma treatment: Atmospheric or vacuum plasma treatments using nitrogen or argon induce partial defluorination and incorporate oxygen and nitrogen functional groups
A third approach bypasses surface preparation entirely. Tech-Bond Global's patented polymer bonding process creates crosslinks directly at the molecular level, achieving permanent bonds to PTFE and PTFE-composite materials without surface adhesives or etching chemistry.
Common Misconceptions
A few persistent misunderstandings follow PTFE across industries — and each one has a straightforward correction.
"Teflon and PTFE are different materials." They aren't. Teflon is a brand name for PTFE, owned by Chemours. All Teflon is PTFE, but not all PTFE carries the Teflon label.
"PTFE works in any chemical environment." Chemical resistance is near-universal, but that's only part of the evaluation. Mechanical load, sealing dynamics, and wear requirements matter too. Dynamic seals under high pressure may need filled PTFE grades or alternative materials like PEEK.
"All PTFE grades perform the same." Virgin and filled grades differ significantly in mechanical strength and wear resistance. A 60% bronze-filled PTFE behaves very differently than unfilled virgin PTFE — treating them as interchangeable leads to premature failure.
Frequently Asked Questions
What is PTFE?
PTFE (polytetrafluoroethylene) is a synthetic fluoropolymer composed entirely of carbon and fluorine atoms, known for near-universal chemical resistance, extremely low friction (0.05–0.10 coefficient), wide temperature tolerance (−200°C to +260°C), and exceptional electrical insulating properties. It's used across industries from cookware coatings to aerospace wiring and industrial sealing systems.
Is PTFE the same as Teflon?
Teflon is a registered brand name (originally by DuPont, now Chemours) for PTFE-based compositions. All Teflon is PTFE, but not all PTFE is Teflon-branded. Other manufacturers produce PTFE under different trade names, and the generic material designation PTFE is used universally in engineering specifications.
What is the operating temperature range of PTFE?
PTFE operates continuously from −200°C to +260°C, making it suitable for both cryogenic and high-heat applications. Degradation begins above 260°C. Pyrolysis with toxic gas emission occurs above 400°C: it must never be overheated beyond this point.
What is the difference between virgin PTFE and filled PTFE?
Virgin PTFE contains no additives, offering maximum chemical resistance and FDA compliance but limited mechanical strength and wear resistance. Filled grades — glass, bronze, carbon, or stainless steel — improve compressive strength, wear resistance, and creep resistance by 200–400% while retaining PTFE's core thermal and chemical properties.
What are the main limitations of PTFE?
PTFE has three key weaknesses:
- Cold flow (creep) under sustained load — compensated with spring washers or filled grades
- Poor elastic recovery, making virgin PTFE unsuitable for pressure-cycling dynamic seals
- Bond resistance to conventional adhesives without surface treatment or molecular bonding
Can PTFE be bonded or glued to other materials?
PTFE's chemically inert, non-polar surface resists conventional adhesives, so traditional bonding requires aggressive pre-treatments like sodium-naphthalene etching or plasma treatment. Molecular-level bonding processes, such as those developed by Tech-Bond Global, achieve permanent bonds to PTFE without surface adhesives by creating crosslinks at the molecular level.


